Quick Summary
ERP Systems for Pharmaceutical organizations help manage compliance, track batches, prevent expiry wastage, and stop counterfeit drugs. This blog explains how these systems solve daily operational problems faced by drug manufacturers and distributors using simple, practical features.
Table Of Contents
Introduction
The pharmaceutical industry faces many challenges when it comes to managing daily functions, keeping up with compliance and regulations, as well as ensuring medicines reach patients without delays or errors. Many organizations still rely on separate systems and spreadsheets, which leads to data mismatches, missed expiry dates, and audit findings. These issues directly impact revenue, reputation, and patient safety. In this blog post, we explain exactly how ERP Systems for Pharmaceutical organizations fix these specific operational gaps. We will see how Pharmaceutical ERP Solutions provide clear, practical fixes for drug manufacturers and distributors.
Key Takeaways
ERP systems centralize compliance and audit trail data for drug manufacturers.
Batch tracking prevents expiry wastage and enables fast product recalls.
Serial number features stop counterfeit medicines from reaching patients.
Real-time inventory improves supply chain visibility and shortage prediction.
Automated regulatory reporting reduces audit preparation time and errors.
Why Generic ERPs Fail Drug Manufacturers
When manufacturers and businesses use very generic and old software to run pharmaceutical operations, specific problems get ignored. Standard business software is not built to handle batch traceability, expiry date monitoring, or strict quality checks. It treats medicines like regular retail products. This is where ERP for Drug Manufacturers becomes necessary.
The general systems do not have the specific modules needed for Pharmaceutical Quality Control ERP. They are unable to identify a batch that fails stability tests or automatically isolate raw materials that do not meet the standards.
Another major gap is serialization, where standard ERPs do not support track-and-trace requirements mandated by regulators. They also cannot manage chemical potency calculations or validate equipment cleaning logs.
The result is manual efforts, human errors, and unsuccessful audits. Organizations then heavily invest in custom solutions or additional software to cover these gaps. This causes more integration problems.
Without the right features in their Pharmaceutical Manufacturing ERP System, drug manufacturers are more prone to compliance risks and operational inefficiencies that directly impact patient safety.
Check out more on ERP Systems for Pharmaceutical.
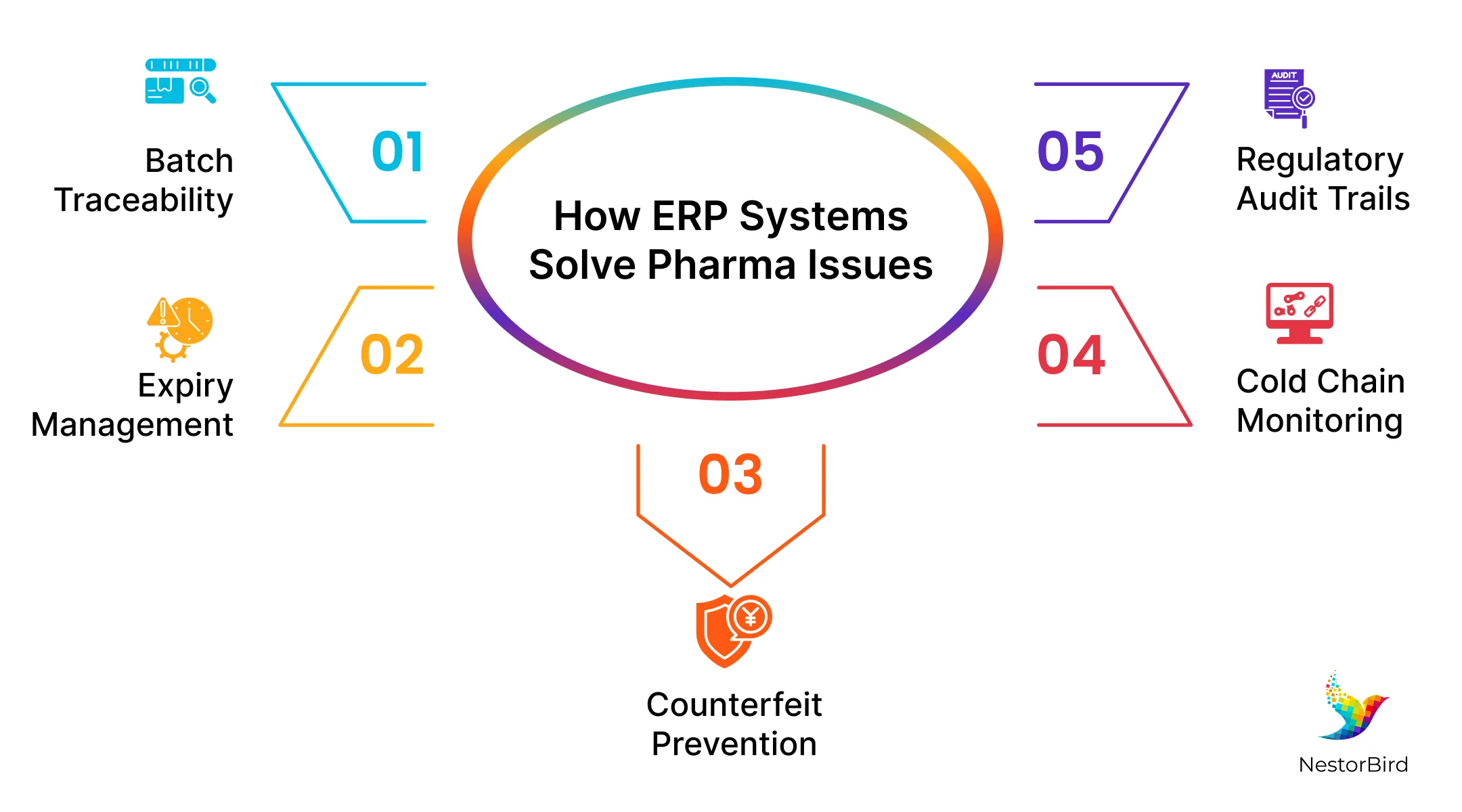
How ERP Systems Solve Issues Faced by Pharmaceutical Organizations
When businesses use and implement the right and best suitable ERP Systems for their functions, specific operational problems get solved directly.
1. Expiry & Wastage
Pharmaceutical Inventory Management ERP changes how stock is handled. Instead of moving old stock first, the system forces First Expiry, First Out automatically. Warehouse staff receive clear instructions on which batch to pick.
The Pharma ERP Software identifies batches that are nearing their expiration date for rapid disposal or sale at a discount. In case of a quality issue, the ERP for Pharma Regulatory Compliance provides a one-click option for batch recall. The system accurately tracks the distribution of every affected batch.
2. Regulatory Fines & Audit Fatigue
Regulators demand complete data transparency. Manual logbooks and spreadsheets fail during inspections. ERP for Pharma Regulatory Compliance creates automated audit trails. Every data entry, edit, or deletion is recorded with timestamps and user IDs. This meets 21 CFR Part 11 requirements for electronic records.
ERP for Pharmaceutical Compliance It produces real-time reports for regulatory submissions, where the data is sourced directly from production, QC, and warehouse logs. The auditors can also access accurate, unmodified information. This cuts down on inspections, eliminates manual report creation, and avoids significant non-compliance penalties.
3. Supply Chain Cold Chain Breaks
Temperature variations during transit can spoil sensitive biologics and vaccines. Pharmaceutical Supply Chain Management ERP integrates with temperature loggers and IoT sensors. If a shipment exceeds the set temperature limits, the system sends immediate alerts. Logistics teams can catch shipments before they are delivered.
The ERP also ensures that carriers comply with cold chain standards before shipping. ERP Solutions for Pharma Distribution ensures that only compliant shipments reach patients. All temperature data is stored in one central location and is available for regulatory review.
This is How ERP Manage Supply Chain for Pharma Industry.
4. Data Silos
Research, manufacturing, and sales teams often work in separate software systems. Information gets stuck between departments. Pharmaceutical Data Management ERP connects all functions on a single platform. Formulation scientists see real-time raw material availability. Production planners access demand forecasts directly from sales. Warehouse teams receive manufacturing completion alerts instantly.
ERP for Pharmaceutical Industry removes any duplicate data entry and manual reconciliation. Decision-makers view consolidated reports instead of merging spreadsheets. This unified approach speeds up batch release, improves forecast accuracy, and removes operational blind spots entirely.
Choose to Improve Batch Processing With Pharma ERP Software.
Protecting Patients and Improving Supply Planning
The presence of counterfeit medicines in the supply chain is a major risk for any drug manufacturer. ERP for Pharma Risk Management addresses this by assigning a unique serial number to each saleable unit. As products move from the factory to the pharmacy, each scan checks for authenticity. If a fake serial number is scanned, the system flags it right away. This protects patients and shields the company from liability.
Moreover, the same data can help forecast shortages. ERP to Improve Pharma Efficiency uses real-time consumption data from hospitals to automate restocking. Stock levels are continuously monitored, and reorders are automatically initiated when inventory is low.
Know the Importance of Business Intelligence in Pharma Manufacturing.
Conclusion
The pharmaceutical industry cannot afford errors in compliance, inventory, or supply chain visibility. Using separate systems and manual processes creates too many risks. Pharmaceutical ERP Solutions provide one unified platform to manage manufacturing, quality, distribution, and regulatory reporting. When looking for ERP Systems for Pharmaceutical needs, choose a partner who understands industry depth. NestorBird builds compliance-ready platforms specifically for regulated industries. Our solutions are designed to handle complex pharma operations from day one without lengthy customizations.
Frequently Asked Questions
The main purpose is to manage compliance, traceability, and inventory in one system. It helps drug manufacturers meet regulatory standards and reduce operational errors.
It creates automatic audit trails for all data entries and edits. This meets 21 CFR Part 11 requirements and generates real-time reports for regulator inspections without manual work.
Generic ERP lacks batch tracking, potency calculations, and serialization features. Pharmaceutical Manufacturing ERP System includes these specific modules required for drug production and compliance.